  
The fourth full-filled hybridized orbital holds the lone pair of electrons.Three half-filled SP3 orbitals of nitrogen form a bond with three hydrogen atoms.During hybridization, one s orbital and 3p orbitals hybridize to form four hybrid orbitals of equal energy levels, making SP3 hybridization.Nitrogen in its ground state has the configuration 1S22S22P3.In the ammonia Lewis structure, the hydrogen atoms are the outside atoms, and each one of them cannot keep more than two electrons in its last shell. Therefore, we will draw the remaining electron pair on the central nitrogen atom.Įlectronic Configuration of Nitrogen in Ammonia Now we have 2 valence electrons to distribute. Out of eight electrons, six will be used in pairs between atoms. In the figure, one electron pair between two atoms is equivalent to one line. We need to distribute the 8 valence electrons. VEs = VEs in three hydrogen atoms + VE in one nitrogen atom The table below shows the electronic configuration and valence electrons in hydrogen and nitrogen. 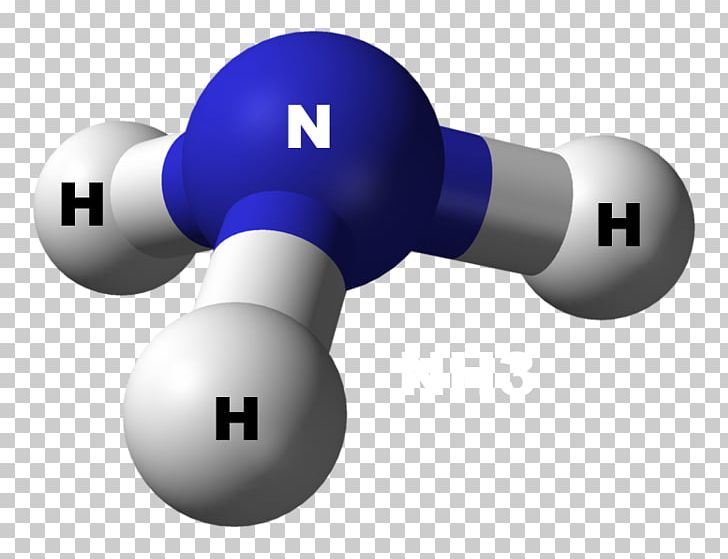
Step-1: Count the Valence Electrons of AtomsĪmmonia contains two elements hydrogen and nitrogen. Hydrogen is a Group IA element and has only one electron in its valence shell. Nitrogen is a group VA element in the periodic table and contains five electrons in its last shell. The following are the steps to construct the ammonia Lewis Structure. Step by Step Construction of Lewis Structure The NH3 molecule is held together by the strong N–H nitrogen–hydrogen single covalent bonds by sharing electrons. Three hydrogen atoms and one nitrogen atom combine to form NH3 so that the hydrogen atoms are electronically like helium and the nitrogen atom becomes like neon. #Nh3 molecular geometry fullEach hydrogen atom is one electron short of a noble gas structure (full shell), and nitrogen is three electrons short of a full outer shell (of eight). Three hydrogen atoms share their electrons with the five outer electrons of nitrogen, essentially giving all four atoms full outer shells. The compound ammonia is formed when three hydrogen atoms interact with one nitrogen atom. Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Strong intermolecular hydrogen bonding makes it highly associated.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians. #Nh3 molecular geometry how to
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
